Abstract:
Introduction: Asthma and other pediatric respiratory disorders are among the most prevalent chronic conditions in childhood, frequently necessitating long-term pharmacotherapy. Although agents such as leukotriene receptor antagonists, corticosteroids, and beta-2 agonists are widely regarded as safe and effective, accumulating post-marketing evidence has raised concerns regarding potential neuropsychiatric adverse events. Reported manifestation including: mood disturbances, sleep disorders, irritability, agitation, and hallucinations ,may be underrecognized in children due to developmental and diagnostic challenges.
Despite regulatory warnings for selected therapies, comprehensive comparative safety assessments across commonly prescribed pediatric respiratory medications remain limited. Therefore, we conducted a national pharmacovigilance disproportionality analysis to systematically evaluate and compare neuropsychiatric safety signals associated with these agents in the pediatric population.
Methods: We conducted a retrospective pharmacovigilance disproportionality analysis using FAERS to detect abnormal neuropsychiatric safety signals for commonly used respiratory drugs. Neuropsychiatric outcomes were agitation, irritability, sleep disturbances, mood changes, and hallucinations. Exposures included montelukast, inhaled corticosteroids (fluticasone, budesonide), systemic corticosteroids (methylprednisolone, prednisone), and beta-2 agonists (albuterol, salmeterol, levalbuterol, salmeterol). Signals were assessed using Reporting Odds Ratios (RORs).
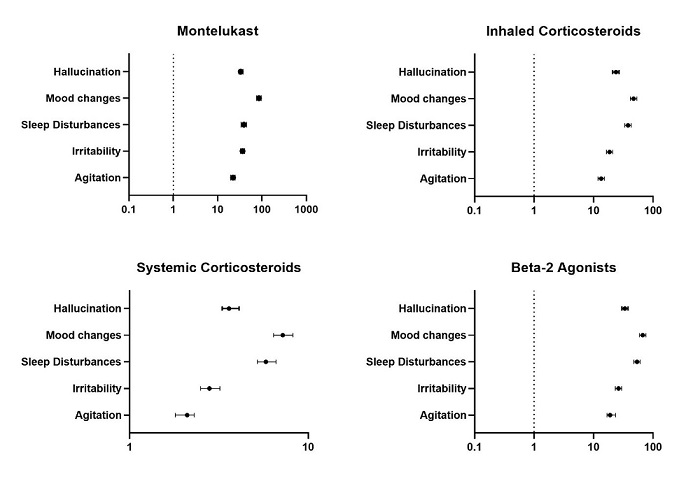
Results: We analyzed 32,679,695 drug-related adverse events. 27,113 reports were related to neuropsychiatric outcomes among the pediatric population. Among the evaluated agents, montelukast showed the strongest signals across all outcomes, particularly for mood changes (ROR 85.15, 95% CI 75.55–95.97), followed by sleep disturbances (ROR 38.89, 95% CI 33.72–44.85), irritability (ROR 36.36, 95% CI 32.50–40.69), hallucinations (ROR 32.89, 95% CI 28.98–37.33), and agitation (ROR 22.05, 95% CI 19.59–24.83). Beta-2 agonists highest signals was for mood changes (ROR 66.88, 95% CI 59.11–75.68) and sleep disturbances (ROR 53.65, 95% CI 47.46–60.64). For inhaled corticosteroids, signals remained substantial, especially for mood changes (ROR 47.07, 95% CI 41.51–53.38) and sleep disturbances (ROR 37.82, 95% CI 33.38–42.84. In contrast, systemic corticosteroids demonstrated weaker but still significant associations, with the highest reporting for mood changes (ROR 7.25, 95% CI 6.44–8.18) and sleep disturbances (ROR 5.83, 95% CI 5.17–6.56).
Conclusion: In this large scale national post marketing analysis, significant neuropsychiatric safety signals were identified across multiple pediatric respiratory therapies. Montelukast demonstrated the most robust and consistent disproportionality signals across all evaluated outcomes. Notably, beta-2 agonists and inhaled corticosteroids also exhibited substantial associations, while systemic corticosteroids showed comparatively lower but statistically significant signals.
These findings reinforce growing safety concerns and emphasize the necessity of vigilant clinical monitoring, individualized risk/benefit assessment, and informed caregiver counseling. Prospective and mechanistic studies are warranted to clarify causality and identify vulnerable subgroups, thereby optimizing the safe use of respiratory pharmacotherapies in children.
Biography:
I am a medical intern at Prince Hamzah Hospital and a graduate of Mutah University Faculty of Medicine. My academic focus centers on clinical research and advanced pharmacovigilance methodologies, with particular interest in post marketing safety surveillance and large scale database analyses. I am committed to strengthening evidence-based prescribing practices through rigorous evaluation of adverse drug reactions. My current research explores neuropsychiatric safety signals associated with commonly prescribed respiratory therapies in pediatric populations, aiming to inform safer therapeutic decision making and optimize patient outcomes.
Copyright 2024 Mathews International LLC All Rights Reserved